journal entry
Dec 20
2017
How to Shoot for the Moon
Whatever did happen to the Cancer Moonshot Initiative?! This initiative was meant to accelerate cancer research and help scientists and clinicians make ten years’ worth of progress toward a cure in only five. But it recently occurred to me I didn’t really know much else about it or what happened after President Obama introduced it during his last State of the Union address two years ago. So, as a true scientist, I decided to dissect its goals and explore how it has progressed so far. Spoiler alert— things are moving slowly. But don’t become disenchanted just yet. It’s for all the right reasons.
When I first started reading about the initiative, I realized I knew very little about the intricacies of the legislative process. First, I was surprised to find out how emotionally driven the process can be. Vice President Joe Biden’s passionate and monumental role in spearheading the initiative was definitely shaped by the passing of his son, Beau, from brain cancer. It also helped rally bipartisan efforts in support of the Moonshot. Second, I didn’t realize the timeline of events leading up to the passage of the bill took almost a year. With all this support, why did it take so long?
It all started to make sense when I noticed the complex and intricate work that went into creating and shaping the initiative. After the Moonshot was introduced in January, 2016, while a special task force was formed to assist in putting together the specific aims of the initiative, House and Senate committees discussed the logistics of funding; they also argued about other research initiatives and what changes to include. In April, 2016, four months after the Moonshot Initiative was introduced, the Blue Ribbon Panel (BRP) was created. Comprised of scientific experts in the field of cancer healthcare and research, the panel included seven specialized groups. These specialized groups were charged with methodically discussing and recommending the aims of the initiative. In September, 2016, the BRP published its report; three months later, Congress passed the initiative as part of the 21st Century Cures Act. With all these different committees, I am now actually surprised it took less than a year! Still the plan seemed a bit too ambitious to me.

Knowing how complex and unpredictable biomedical research is, I was skeptical at first about how this five-year plan was going to play out. Curing cancer is actually much more complicated than the moon landing. Figuring out how to land on the moon was like completing a 1000-piece jigsaw puzzle-- it’s hard, but there are very clear and methodical steps to follow. Figuring out how to cure cancer, on the other hand, is like trying to solve a 1000-piece jigsaw puzzle while more than half the pieces are missing and the complete picture is blurred. Besides, cancer is not a single disease. Tumors are unique to each organ and individual, so we are actually trying to solve multiple different incomplete jigsaw puzzles.
Close examination of the 10 recommendations or goals in the BRP’s report, however, showed me just how we can, indeed, achieve such progress in half the time originally identified as necessary. Phrases like 'data ecosystem' and 'new cancer technologies' jumped right out at me and I started seeing a similar pattern in the recommendations. It dawned on me they can be divided along two categories, either clinical research and progress or basic and translational research.

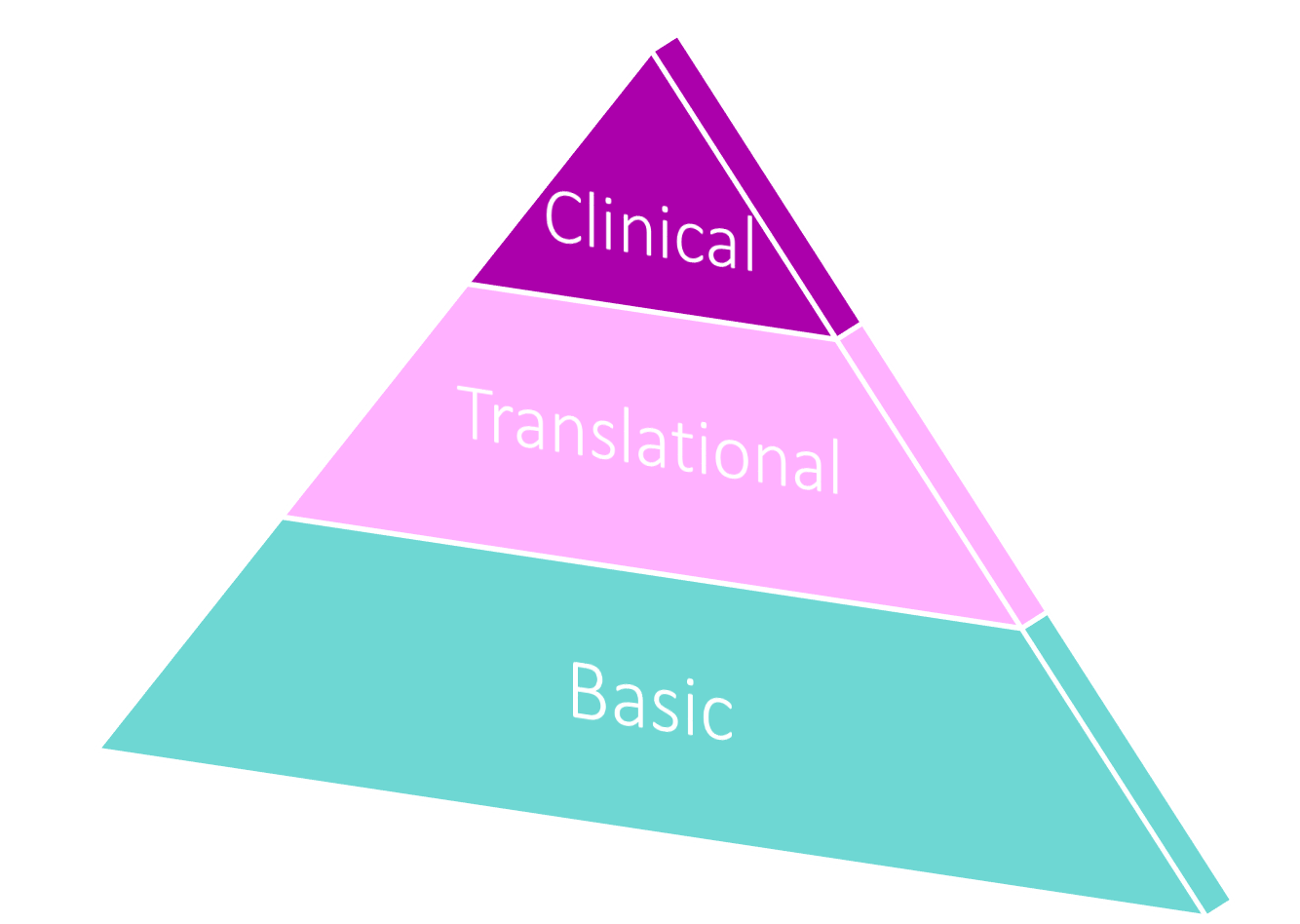
Now before I lose you with that jargon, think of biomedical research as a pyramid. At its base is basic research, or discovery science, where experiments are aimed at discovering and improving our fundamental understanding of nature. Translational research makes up the middle layer. It uses these basic scientific discoveries to develop novel technologies and drugs to cure and manage disease. The top of the pyramid is clinical research and progress, where successful technologies and drugs are tested on patients, some of which eventually progress into clinical practice. You can easily imagine it takes a lot more time to start building a pyramid from the base up than starting from the middle or top.
Consequently, for this time-sensitive initiative, these recommendations lean more toward clinical research and progress and less on discovery. They are purposely not building a pyramid from scratch here. Specifically, the clinical research and progress category focuses on collecting, gathering, reinterpreting, and disseminating readily available pieces of the puzzle and connecting them in order to create a more complete portion of the picture. Here, scientists and clinicians will use the vast amount of clinical information to mine past patient data and build a national cancer data ecosystem.
Assembling this information into a structured, accessible, and clear database will help patients, clinicians, and scientists communicate better and predict outcomes to standard treatments. Along these lines, scientists will also develop a 3D cancer atlas, an interactive live puzzle that shows the progress of tumors from small legions to metastasis. It will help scientists observe and assess the environment in which tumors grow and give clinicians context as to how treatment should best be pursued. These clinical research and progress goals won’t necessarily lead to new cures but they will significantly improve patient experiences, prevention measures, and clinical outcomes in half the time.
The goals of the basic and translational research category, however, focus on actually finding new (basic) puzzle pieces and linking them to the ones we have found so far (translational). Essentially, they aim at discovering new drivers of cancer in the hopes of eventually targeting them to stop tumors from forming or resisting treatment. These drivers can also be used as “tags” or biomarkers that help evaluate our risk of certain cancers and improve prevention and early detection measures. Biomarkers include defects or mutations in specific essential genes that can eventually lead to the development of certain tumors. One of the very few established cancer biomarkers is a defective BRCA gene, which can be genetically tested to predict a person’s chances of developing hereditary breast, ovarian, and prostate cancers, among others. Nonetheless, how will these tags or drivers be targeted to eliminate cancer? Well, since it’s the only treatment option with its very own aim, the BRP strongly recommends immunotherapy. This approach, which uses our own immune systems to specifically attack and kill cancer cells, has been making accelerated advances for treatments of many types of cancers recently. Unlike chemotherapy and radiation, it can be a personalized and cancer-specific strategy that will lead to better prognoses and fewer side effects. Both academic scientists and biopharmaceutical companies are rallying around this promising treatment as the National Institute of Health has recently announced a Partnership for Accelerating Cancer Therapies (PACT) with 11 leading biopharmaceutical companies to develop new cancer immunotherapies. Grouped together, these aims and partnerships demonstrate the focused and comprehensive vision of the initiative and form a clear guide to shooting for an elusive moon.
This “year in a life” of the Cancer Moonshot Initiative chronicles its launch and foundation. The actual groundwork is happening now. Scientists and clinicians have started working on projects supported by the initiative’s goals and funds, and will continue to do so through 2023. We cannot completely predict the level of progress we’ll achieve by then but the cancer field seems to have etched a clear path toward which it is heading, making it a terrific time for progress in cancer prevention, detection, and care. Personally, I am excited to explore this path, collecting accessible pieces of the puzzle and sharing them with you along the way.




